Fighting Cancer and Infectious Diseases


KUPANDO is a pioneering biotech company developing disruptive therapies to address critical unmet medical needs with significant market potential in cancer and infectious diseases.
Kupando’s disruptive approach harnesses the power of innate immune stimulation and induction of trained innate immunity by dual Toll-Like Receptor (TLR) agonists.
Our lead candidate, KUP101, is a differentiated and competitive dual TLR agonist with a robust preclinical profile and a clear path to the clinic with a pipeline of multiple programs targeting oncology and infectious disease indications.

OUR APPROACH
DRUG CANDIDATES: TLR4/7 AGONISTS
Kupando is pioneering a new era in cancer therapy and infectious diseases management with its innovative dual TLR agonist through the power of innate immune activation and induction of trained innate immunity.
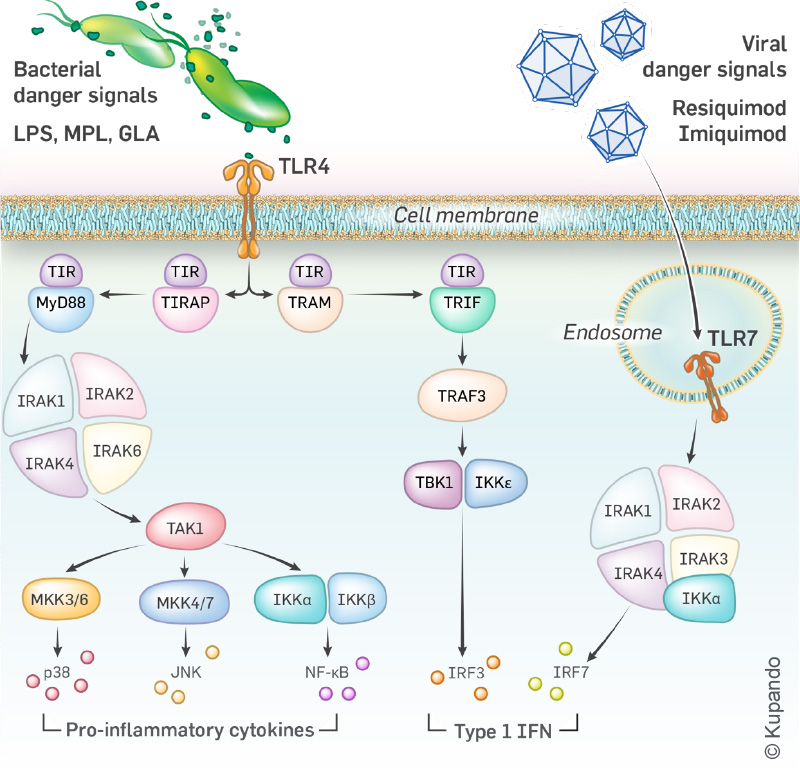
Toll-Like Receptors (TLRs) are key components of the immune system responsible for detection of cancer and infection signature molecules. When they detect a threat, such as a virus or a tumor cell, they activate the immune system to fight back. TLR4 and TLR7 are two important TLRs, each with unique roles in recognizing and responding to different threats.
Kupando develops innovative drug candidates – TLR 4/7 agonists – which activate both TLR4 and TLR7 simultaneously, unleashing a powerful immune response, both harnessing innate immunity and inducing trained immunity.
Kupando’s lead compound, KUP101, is formulated to provide a multitude of options for administration routes and applications.

A UNIQUE TOOL TO FIGHT CANCER AND INFECTIOUS DISEASES
Our proprietary drug candidates are ideally suited for the systemic treatment of solid tumors (tissue agnostic), and the prevention and treatment of infectious diseases. Kupando is focusing on critical unmet medical needs, such as treatment-resistant cancers and antimicrobial-resistant infections.
A GAME-CHANGER
Kupando’s drug candidates are first-in-class, differentiated and competitive small molecules with the potential to transform the management of cancer and infectious diseases. The innate immune system is highly conserved. As such, its efficacy treating cancer or treating/preventing infections is tumor/ pathogen agnostic.
Kupando’s lead drug candidate, KUP101, offers hope for patients facing difficult-to-treat conditions. In cancer, it may extend therapeutic options for those who do not respond to checkpoint inhibitors.
KUP101 also provides a promising new approach to combat infectious diseases, especially antimicrobial resistant infections. As a host-directed agent – unlike antibiotics – it has the potential for highly effective therapeutic as well as prophylactic applications, while mitigating the risk of pathogen resistance.
This is a game changer in the management of both these critical diseases. KUP101 is currently the only therapy in development that combines TLR 4 and TL7 agonists.
Proof-of-concept data have been confirmed in multiple animal experiments and independent research units, including use as single agent or in combination with other agents such as checkpoint inhibitors. CTA-enabling work is ongoing for the treatment of solid tumors and the treatment and prevention of infectious diseases including antimicrobial resistant pathogens.
Advantageous features
- TLR4 and TLR7 are validated targets
- High specificity for target
- No relevant off-targets
- Fast intracellular uptake, leading to improved safety
- Combines both TLR4 and TLR7 agonist
- Advantageous administration route (formulated for systemic administration)
- Tumor agnostic, pathogen agnostic (broad immune response including epitope spreading)
- Effective as single agent or in combination (synergistic in combination with checkpoint inhibitors)
- Host-directed agent, no induction of drug resistance when used to treat or prevent infectious diseases
PIPELINE
Kupando is committed to developing innovative therapies that can make a real difference in the lives of patients. Our pipeline focuses on KUP101, a first-in-class dual TLR agonist with the potential to transform the treatment and /or prevention landscape for multiple indications in cancer and infectious diseases.
| Program | Indication | Stage of development | Proprietary/ Partnered |
|---|---|---|---|
| KUP101- Oncology | Skin cancer Cutaneous Squamous Cell Carcinoma (cSCC) | CTA-enabling | Proprietary |
| KUP101- Oncology | Solid tumors (lung, breast) | Pre-clinical | Proprietary |
| KUP101- Infectious Diseases | Anti-Microbial Resistance (AMR) | Pre-clinical | Proprietary - funded by Federal Ministry of Education and Research (Germany) |
| KUP101- Infectious Diseases | Vaccine | Pre-clinical | Undisclosed Partner |

[click image to enlarge]
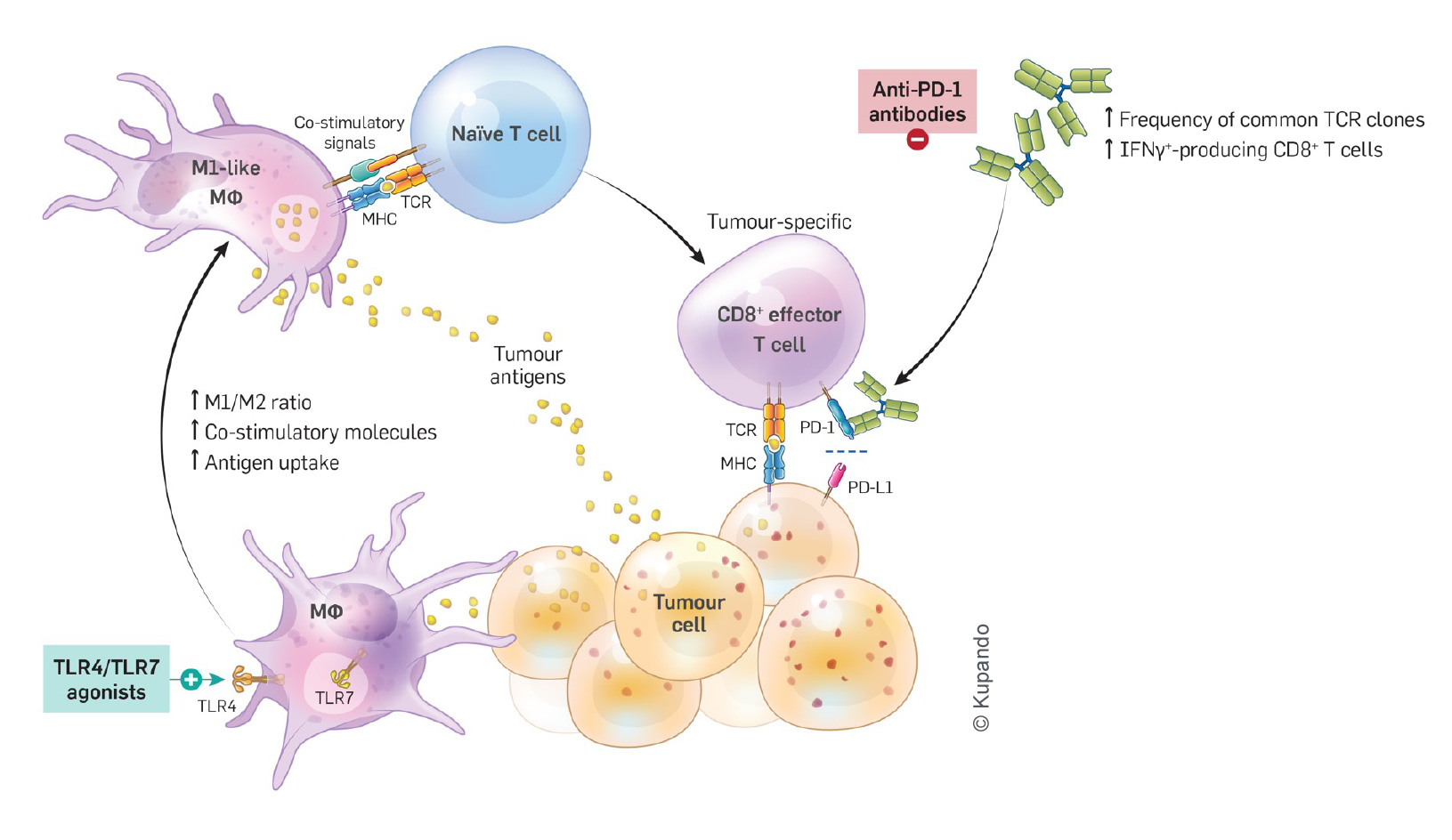
TLR Agonists Activate a Different MoA from Checkpoint Inhibitors — Additive Effect
FIGHTING CANCER BEYOND CHECKPOINT INHIBITION
Checkpoint inhibitors have revolutionized cancer treatment, but they don't work for everyone. Some patients don't respond, and others develop resistance over time. Plus, these treatments can cause serious side effects.
TLR4/7 agonists work differently than checkpoint inhibitors and target different parts of the immune system.
Kupando's lead drug candidate KUP101 has the potential to be used as a standalone treatment for a variety of solid tumors (tissue agnostic), regardless of their location in the body. It can also be used in combination with other drugs to enhance their effectiveness. In vivo studies have shown a synergistic/additive effect with checkpoint inhibitors. KUP101 offers a new hope for patients who haven't been helped by other treatments.

FIGHTING INFECTIOUS DISEASES BEYOND ADAPTIVE IMMUNITY
Kupando’s TLR 4/7 agonists work by activating the innate immune system and inducing trained immunity, leading to a more effective response against infections, and offering a promising new approach to protection from serious infections.
"Superbugs" or Anti-Microbial-Resistant (AMR) infections pose a major threat to global health. Activation of innate immunity and induction of trained immunity offers a promising game-changing approach to treating and preventing these serious infections.



Funding reference number: 16LW0713
Topic: KMUi-Biomedicine-4: Development of a pathogen-agnostic therapy and prophylaxis against AMR by stimulation of the patient's innate immune system (PANTHER)
Project description:
For many years, the employees of KUPANDO GmbH have been working on the development of agonists that bind to toll like receptors and thus lead to the activation of the innate immune system. Toll like receptors are located on the cell surface or inside the cells of immunocompetent cells. It is known that the innate immune system, the oldest defence system in evolution, plays a key role in the defence against infections and in the fight against oncological diseases. In recent years it has also become known that the innate immune system also has a memory, which is referred to as ‘trained immunity’. The importance of this training effect has been demonstrated in infectiology and oncology.
Antimicrobial resistance (AMR) is one of the major challenges in healthcare today and will become even more important in the future. Some scientific studies have shown that the stimulation of TLRs can contribute both in the prophylaxis and therapy of AMR.
Many cellular and animal experiments have shown that the stimulation of TLR4 and TLR7 together (KUP 101) triggers a very broad immune response. This broad immune response has been shown to be effective in the treatment of solid tumors and in the prophylaxis of infectious diseases. The immune response is not pathogen-specific and is therefore broadly applicable. This unique tool is now to be used to contribute to the prevention and treatment of AMR.
The approach differs from others. Instead of attacking the pathogen, such as with antibiotics, the defence system of the host, the patient, is strengthened so that it can successfully defend itself against the infection.
The aim of this project is to show that a safe and effective therapy and prophylaxis of AMR can be developed in vivo with KUP 101. During development, Kupando can draw on the toxicological data and the development of the manufacturing process that have already been carried out for the indication of solid tumors.
Scientific Background
Vertebrates possess two interconnected immune systems: innate and adaptive. Innate immunity, the evolutionarily older system, provides rapid, broad-spectrum defense against pathogens. While historically underappreciated, it is now recognized as a critical target for immunomodulation. Kupando's approach centers on harnessing this powerful system.
TLR 4/7 agonists stimulate innate immunity by activating Toll Like Receptors (TLRs) and therefore provide a unique tool to fight cancer and infectious diseases.
TLRs: The Innate Immune System’s sentinels
TLRs operate to detect signature molecules that herald infection and cancer by activating the innate immune system.
There are 10 known TLRs in the human innate immune system. KUP101 specifically stimulates TLR 4 and 7 (TLR 4/7 agonist).
In primates, TLR 7 is primarily expressed by plasmacytoid dendritic cells and B cells. Active TLR 4 is expressed by many more cell types, including endothelial cells and macrophages.
Activating the Defense System
The innate immune system is like a first responder, quickly reacting to threats. To start a longer-lasting fight, it needs to team up with the adaptive immune system.
If a tumor has limited infiltrating dendritic cells and B cells, a TLR 7 agonist alone may not be a sufficient stimulant. KUP101’s formulation though, which provides additional co-localized TLR 4 agonists, induces local cytokine and chemokine release, especially from the endothelial cells that line the abundant micro vessels that penetrate tumors. In turn, this leads to the recruitment of more dendritic cells and B cells from the blood stream to the tumor, promoting a robust CD8 cytotoxic immune response.
Inducing Trained Immunity
When KUP101 activates TLR 4 and TLR 7, it not only targets innate immunity to elicit a potent and durable immune response, but it also enhances the innate immune system’s responsiveness against future threats. This is called ‘trained immunity’.
Trained immunity can be described as a functional state of the innate immunity, which is defined by long-term epigenetic and metabolic reprogramming of innate immune cells. This “training” effect leads to enhanced responsiveness to subsequent challenges, to any second signal, even if it is new to the host. Trained immunity provides therefore the means for a pathogen or tissue agnostic treatment.

[click image to enlarge]
KUP101: A New Kind of Immunotherapy
Both TLR 4 and TLR 7 are validated therapeutic targets, exemplified by their use in existing vaccine adjuvants and cancer therapies. Kupando's KUP101 takes this one step further by not only activating these targets but also activating trained immunity, teaching the body’s first line of defense to fight off future threats more effectively.
Unlike traditional immunotherapies that focus on the adaptive immune system, KUP101 works by enhancing the innate immune system, causing changes in innate immune cells, like epigenetic modifications and metabolic reprogramming, making them more responsive.
This unique approach allows KUP101 to offer a new pathogen or tissue agnostic way to fight cancer and infectious diseases, potentially providing longer-lasting protection and overcoming some of the limitations of existing treatments.
Publications
Kupando’s drug development is supported by scientific research, see below a short list of scientific publications. For additional information or publications by our team or our collaborators, please talk to us.
December 2025
Trained Immunity: RoadMap for drug discovery and development
van Puffelen et al.
eLife
October 2022
A Dual Adjuvant System for Intranasal Boosting of Local and Systemic Immunity for Influenza Vaccination
Sato-Kaneko et al.
Vaccines
June 2020
A Novel Synthetic Dual Agonistic Liposomal TLR4/7 Adjuvant Promotes Broad Immune Responses in an Influenza Vaccine With Minimal Reactogenicity
Sato-Kaneko et al.
frontiers in Immunology
June 2018
Induction of oligoclonal CD8 T cell responses against pulmonary metastatic cancer by a phospholipid-conjugated TLR7 agonist
Hosoya et al.
PNAS
Sept 2017
Combination immunotherapy with TLR agonists and checkpoint inhibitors suppresses head and neck cancer
Sato-Kaneko et al.
JCI Insight
July 2017
Synthetic Toll-Like Receptor 4 (TLR4) and TLR7 Ligands Work Additively via MyD88 To Induce Protective Antiviral Immunity in Mice
Goff et al.
Journal of Virology;
American Society for Microbiology
Partnering
Partnering Proposition
At Kupando, we're pioneering a new era in cancer and infectious disease prevention and treatment through the power of innate immune activation and trained innate immunity. Our innovative dual TLR agonist approach has the potential to transform the way we fight these diseases.
We're seeking partners who share our vision for transforming patient care. We are currently open to co-development partnerships in other solid tumors, either as single agent or combination therapies.
Partner with Kupando and help us develop innovative therapies for cancer and infectious diseases.
Contact us to find out more.

Our existing Collaborations
Ruhr-Universität Bochum (Skin Cancer):
Partnering with the University of Bochum for over 2 years, we're advancing skin cancer research. Our collaboration focuses on understanding TLR4/TLR7 expression in healthy volunteers and skin cancer patients. Prof. Stockfleth acts as Principal Investigator for our clinical trials.
Infectious Disease Collaboration:
We are collaborating with a partner on innovative vaccine research. This partnership combines our expertise in innate immune activation and induction of trained innate immunity by dual TLR agonists with their knowledge in vaccine development to develop new ways to fight infectious diseases.


About Kupando
Our Name
Our Company Name “Kupando” derives from the Swahili word “kupanda”, which means to rise or ascend. We chose this name because it reflects our hope for the patients who may benefit from our drug candidates.
Our goal is to create better drugs in oncology and for the fight against infectious diseases including those common in developing countries. Our focus on AMR for example, a global health crisis, will have a significant impact on communities in regions like Kenya, where Swahili is spoken.
This is why we chose the ascending crown crane as our Company Bird. A symbol of good luck and the guardian of the health of people and cattle, it is revered in many cultures in the Sahel region of Africa (south of the Sahara). It represents our commitment to improving global health.
The picture of our Company Bird was taken by our CEO, Johanna Holldack during a trip to Kenya, where she came up with the concept behind Kupando.

Management
Kupando is led by a management team with a wealth of international biotech and pharma experience gained in both Europe and the US in the fields of immunology, infectious diseases and oncology.
With over 100 drug development projects and a huge variety of transactions under its belt, our team brings extensive expertise in business and drug development including in manufacturing, non-clinical and clinical development of vaccines and drugs, as well as regulatory pathways in Europe, the US and Asia.

CEO & Founder
- 30+ years’ experience in drug development:
- Oncology (Proleukin, Eligard, Veregen, TMX-101, etc.)
- Vaccines (Influenza, Rabies, DTaP, etc.) - 4 successful Exits (IPO and trade sales: MediGene, Borean Pharma, Telormedix, Amal Therapeutics)

CFO
- Corporate Finance & Investor Relations
- Managed two IPOs and several M&A transactions
- Worked as top Management Consultant and Start-up Coach
- 20 years’ experience in the Biotech Industry
- Business Economist by training

Strategic Marketing & Business Development
- 30+ years’ experience across pharma, biotech and venture
- Product development strategy, business development, strategic marketing
- Global Head, VP Strategic Marketing at Novo Nordisk, Telormedix
- Venture Partner, Aravis

Head of Operations
- 35+ years’ experience in drug development (Sermino, Cabaser, Reboxetine, Xadago, TMX-101)
- Expert in CMC, Preclinical Development and Toxicology
- VP of Development at Gain Therapeutics listed on NASDAQ
- 2 successful Exits as Co-founder of Telormedix and Head of Development at Inositec

Head of Clinical Operations & Project Management
- 15+ years’ experience in Clinical Operations
- Director Clinical Operations at MiGenTra and MOLOGEN
- Head of Clinical Project Management at Cardior Pharmaceuticals
- Clinical Trial Management
(early phase oncology/hematology)
at NOXXON Pharma
Advisory Board
Kupando is backed by an industry approved advisory board.

Chairman
- Chairperson of the Boards of Genespire, Captain T Cell, HHC Medical, member of the Board of Trustees at Deutsches Rheuma Forschungszentrum, Member of Volt Europe to support the professionalization of the party
- Managed the scale-up of several biotech companies, including Evotec, uniQure, Hookipa
- Has overseen the approval of the first Western gene therapy product, and led multiple stock market listings on Nasdaq, SIX SE and Frankfurt SE

Deputy Chairman
- Head of Asset Leadership and member of the R&D Leadership Team at AbbVie (USA)
- Held global R&D leadership positions at Takeda, Shire, Abbott, Astellas, Hoechst Marion Roussel and others
- Has overseen the design and conduct of several key global development programs and successful product registrations

Board member representing Remiges Ventures
- Principal at Remiges Ventures
- Lead and contributed toward many drug discovery projects including oncology and cardiovascular areas
- Has worked for Novatis, Boheringer Ingelheim, Mochida Pharma, RaQualia Pharma

Board member representing a private foundation
- CEO & Founder of LifeCare Partners and kineo finance
- Founded, built and managed BioMedPartners, InterPharmaLInk and worked for McKinsey and Novartis
- Led 80+ investments in life science companies

Board member representing Brandenburg Kapital
- Investment Director Life Sciences, Brandenburg Kapital GmbH
- 15+ years’ industry experience in drug development, diagnostics, and life science tools
- Held BD and M&A positions at Sartorius AG, Thermo Fisher Scientific, and Mologen AG
Scientific Advisers
Kupando's work is inspired by pioneering research conducted at the University of California San Diego (UCSD) in the laboratories of Prof. Dennis Carson, whose work with TLR4/7 agonists laid important groundwork in this field.

Inventor and Scientific Advisor
- Currently working on adjuvant discovery and vaccine research
- Played key role in founding Dynavax, IDEC, Telormedix, Vical and others
- About 600 papers published
- Professor Emeritus, Moores UCSD Cancer Center
- Member of the National Academy of Science

Scientific Advisor
- Researching on the molecular mechanisms of innate immunity and inflammation.
- Most cited immunologist in Europe
- President, Fondazione Humanitas per la Ricerca
- Emeritus Scientific Director, Istituto Clinico Humanitas
- Emeritus Professor, Humanitas University

Clinical and Scientific Advisor
- Currently researching in the field of dermatology, oncology and in particular skin cancer
- Director of the University Department of Dermatology at St. Josef-Hospital in Bochum
- Former European President of Skin Care in Organ Transplant Patients Network (SCOPE)

Investors

Press Releases

Kupando Secures Additional €10 Million in Series A Financing to Advance Novel Immuno-Oncology and Infectious Diseases Programs
Schönefeld, Germany, 18 March 2026 – Kupando Secures Additional €10 Million in Series A Financing to Advance Novel Immuno-Oncology and Infectious Diseases Programs

Kupando Appoints Biotech Industry Veteran Jörn Aldag as Chair to Accelerate Development of TLR 4/7 Agonist Platform
Schönefeld, Germany, 3 February 2026 – Kupando, a pioneering biopharmaceutical company developing a TLR 4/7 agonist that stimulates innate immunity and induces trained immunity for use in oncology and infectious diseases, today announced the appointment of Jörn Aldag

Kupando raises €13 million in Series A funding round
Schönefeld/Berlin, 26 September 2022 – Kupando, a pioneering biopharmaceutical company developing TLR 4/7 agonists that stimulate innate immunity for use in oncology and infectious diseases, announces today the closing of its Series A funding round which raised €13 million.


Meet us at
HTGF Family Day
11-12 May 2026
Berlin, Germany
Johanna Holldack, CEO & Founder attending
Thomas Dümmler, Head of Clinical Operations & PM attending
PHARMA PARTNERING SUMMIT
20-21 May 2026
Basel, Switzerland
Johanna Holldack, CEO & Founder attending
BIO:CAP EUROPE
10 Juni 2026
Berlin, Germany
Johanna Holldack, CEO & Founder attending
World Vaccine Congress I EUROPE
19-21 October 2026
Amsterdam, The Netherlands
Johanna Holldack, CEO & Founder attending
Jelmer van Puffelen, Scientist Immunology attending
BIO-EUROPE
9-11 November 2026
Cologne, Germany
Johanna Holldack, CEO & Founder attending

Contact
Kupando GmbH
Willy-Brandt-Platz 2
12529 Schönefeld
Germany
E: contact@kupando.com
www.kupando.com
Fighting Cancer and Infectious Diseases